
So although energy was conserved in the transfer (the first law), the entropy of the universe has increased in becoming more disordered (the second law) and consequently the availability of energy for doing work has decreased. We know that the formula for change in entropy is S Q T ( J K) Plugging in the correct values for Q and T, we. 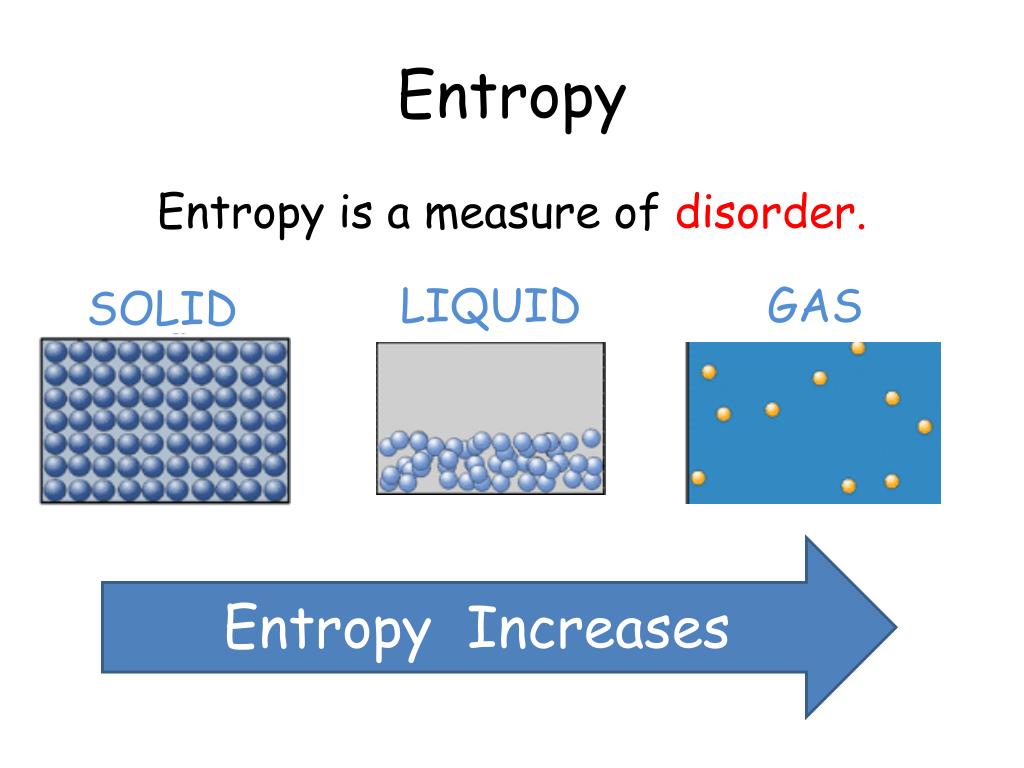 
So as a quick summary, when trying to figure out the change in entropy, we need to consider the number of available microstates. But the process of increasing the disorder has removed the possibility that the energy that was transferred from A to B can be used for any other purpose - for example, work cannot be extracted from the energy by operating a heat engine between the two reservoirs of different temperatures. Step 2: Use the formula for change in entropy to calculate the change in entropy. Therefore, for the change in the entropy, if S1 is greater than S2, we're subtracting a larger number from a smaller number, which means the change in entropy for this reaction, delta S, will be negative. So the disorder of the system has increased - and we say the entropy has increased. When they are put in thermal contact, energy flows from the higher average energy system to the lower average energy system to make the energy of the combined system more uniformly distributed - ie, less ordered. The sense of order is associated with the atoms of system A and the atoms of system B being separated by average energy per atom - those of A being the higher energy atoms if system A is at a higher temperature. When two objects are at different temperatures, the combined systems represent a higher sense of order than when they are in equilibrium with each other. Natural processes will always proceed in the direction that increases the disorder of a system. That disorder can be represented in terms of energy that is not available to be used. Entropy of the universe always increases Entropy is a measure of the disorder of a system. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Use the information below to generate a citation. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: 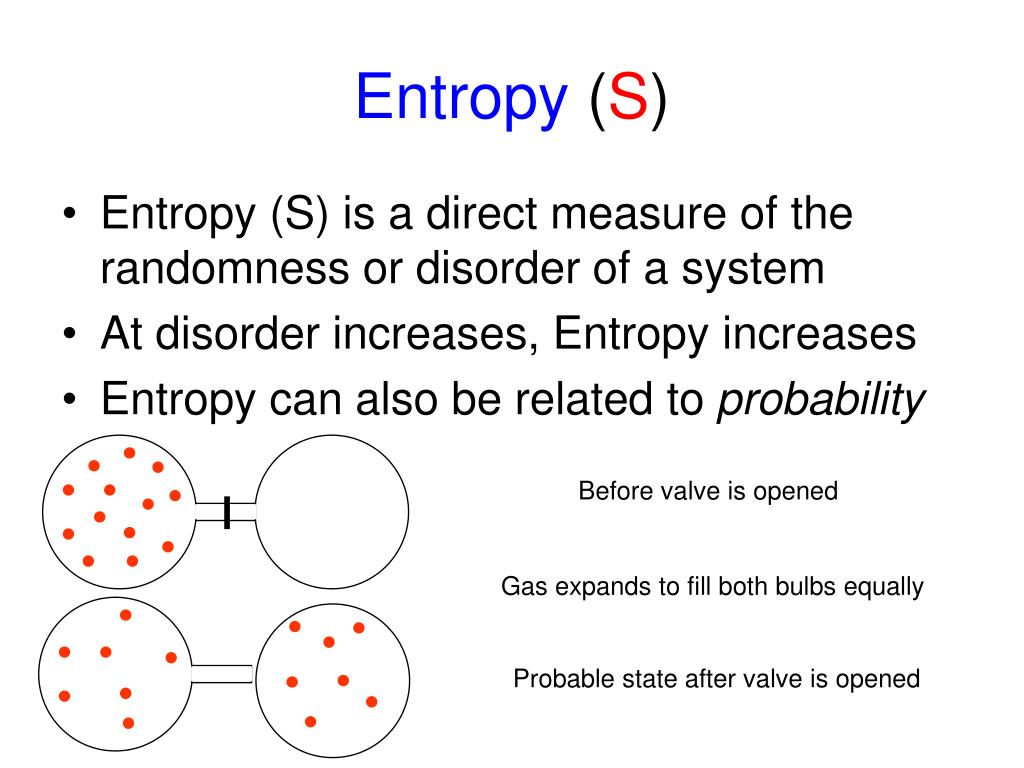
If you are redistributing all or part of this book in a print format, In a chemical reaction, the change in entropy can also be attributed to the rearrangement of atoms or ions from one pattern to another. Thus, the greater the disorderliness in an isolated system, the higher is the entropy. Want to cite, share, or modify this book? This book uses the Hence, change in entropy does not differ with the nature of the processes either reversible or irreversible. The net change in entropy of the system for the transition is Sometimes, you might find the subscript rev in the Clausius definition. The change in entropy for each step is Δ S i = Q i / T i. Therefore, the entropy change has the units of energy divided by units of temperature. This can be accomplished experimentally by placing the system in thermal contact with a large number of heat reservoirs of varying temperatures T i T i, as illustrated in Figure 4.15. During each step of the transition, the system exchanges heat Δ Q i Δ Q i reversibly at a temperature T i. The temperatures associated with these states are T A T A and T B, T B, respectively. Imagine a system making a transition from state A to B in small, discrete steps. The change in entropy of a system for an arbitrary, reversible transition for which the temperature is not necessarily constant is defined by modifying Δ S = Q / T Δ S = Q / T. Since there is no heat transferred into the cylinder and no other losses, the change in entropy is zero. Science Chemistry Calculate the change in entropy when 50 kJ of energy is transferredreversibly and isothermally as heat to a large block of copper at (a) 0☌, (b) 70☌. The same equation could also be used if we changed from a liquid to a gas phase, since the temperature does not change during that process either. where the numbers 1 and 2 denote the states at the beginning and end of the compression process, s is the entropy, T is the temperature, p is the pressure, and 'ln' denotes the natural logarithm function. Δ S = 16.8 kJ 273 K = 61.5 J/K Δ S = 16.8 kJ 273 K = 61.5 J/Kĭuring a phase change, the temperature is constant, allowing us to use Equation 4.8 to solve this problem.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
